Symmetrel (Amantadine)

Dosages
Symmetrel 100 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | A$2.04 | A$61.18 | |
| 60 | A$1.61 | A$96.33 | |
| 90 | A$1.48 | A$132.78 | |
| 120 | A$1.36 | A$162.73 | |
| 180 | A$1.24 | A$222.61 | |
| 270 | A$1.17 | A$316.34 | |
| 360 | A$1.11 | A$398.35 |
Payment & Delivery
Your order is carefully packed and ships within 24 hours. Here is what a typical package looks like.
Sized like a regular personal letter (approximately 24x11x0.7 cm), with no indication of what is inside.



| Delivery Method | Estimated delivery |
|---|---|
| Express Free for orders over A$390.54 | Estimated delivery to Australia: 4-7 days |
| Standard Free for orders over A$260.36 | Estimated delivery to Australia: 14-21 days |










Discount Coupons
- Australia Day - 26 January 2026 10% AUSDAY10
- ANZAC Day - 25 April 2026 8% ANZAC8
- Boxing Day - 26 December 2026 12% BOXING12
Brand Names
| Country | Brand Names |
|---|---|
 Argentina Argentina | Actison Ampakine Virosol |
 Australia Australia | Antadine |
 Belgium Belgium | Amantan Mantadix |
 Brazil Brazil | Mantidan |
 Canada Canada | Endantadine |
 Czechia Czechia | Amantadol PK-Merz Viregyt-K |
 Denmark Denmark | Virofral |
 Finland Finland | Atarin |
 France France | Mantadix |
 Germany Germany | Adekin Aman Amanta Amantagamma Amixx AMT Cerebramed Contenton Grippin-Merz InfectoFlu Infex PK-Merz tregor Viregyt |
 Greece Greece | Hofcomant PK-Merz |
 Hungary Hungary | PK-Merz Viregyt |
 Italy Italy | Mantadan |
 Malaysia Malaysia | PK-Merz |
 Mexico Mexico | Kinestrel Padiken PK-Merz |
 Poland Poland | Amantix Viregyt K |
 Portugal Portugal | Parkadina PK-Merz Profil |
 Spain Spain | Mantaviral Protexin |
 Sweden Sweden | Virofral |
 United States United States | Symadine |
| Manufacturer | Brand Names |
|---|---|
| Cipla Limited | Amantrel |
Description
Amantadine (Symmetrel) is an antiviral medicine mainly used to treat and prevent influenza A infections. However, its effectiveness has reduced because resistance is now widespread. It is also used to help manage symptoms of Parkinson's disease and medicine-induced movement disorders by increasing dopamine levels in the brain. Amantadine comes in several forms, including tablets and extended-release capsules. Symmetrel is a dopaminergic medicine, which means it can increase the levels of certain chemicals that carry signals in the nervous system, including the brain.
Uses
Amantadine is an antiviral medicine that is 70-90% effective in preventing influenza A infections and can reduce symptoms when taken within 24-48 hours of symptoms starting. While it is comparable to rimantadine and vaccination, its effectiveness drops after 48 hours, and resistance among influenza strains is an increasing concern. Amantadine is mainly used for influenza A.
Symmetrel capsules are used:
- to treat Parkinson's disease by improving muscle control and reducing stiffness, shakiness and shuffling;
- to help treat shingles (herpes zoster) and reduce pain.
Symptoms
Certain signs can help distinguish the causes of influenza-like illness. Nasal congestion and rhinorrhoea are common in most cases but rare in inhalational anthrax.
Fever, chills, fatigue, cough, headache, myalgia, sore throat and rhinorrhoea occur in 64-94% of laboratory-confirmed influenza cases. By contrast, similar symptoms appear in 62-94% of patients with other viral or bacterial infections (excluding inhalational anthrax), although fever is present in only 40-73%. Nausea or vomiting occurs in 12%, abdominal pain in 22%, shortness of breath in 6%, and chest discomfort in 23-35% of these patients. In inhalational anthrax, 60-80% of patients have shortness of breath and chest pain, 80-90% have nausea, and 70% report drenching sweats.
Initial chest X-rays in patients with inhalational anthrax often show mediastinal widening, infiltrates and pleural effusion. While most influenza-like illnesses do not show pneumonia on X-ray, it can occur in young children, older adults, and people with chronic lung disease. Influenza-associated pneumonia affects about 1-5% of adults and more than 20% of older adults infected with influenza.
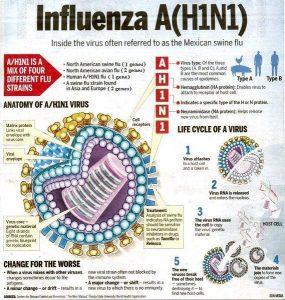
Mechanism of Action
The exact mechanism behind the antiviral activity of amantadine has not been fully established.
Amantadine, like rimantadine, inhibits viral replication by interfering with the influenza A virus M2 protein, an integral membrane protein. The M2 protein of influenza A acts as an ion channel and is essential in at least two parts of virus replication: disassembly of the infecting virus particle and regulation of the ionic environment of the transport pathway. By interfering with the ion channel function of the M2 protein, amantadine inhibits two stages in the influenza A replication cycle. Early in the cycle, amantadine inhibits uncoating of the virus particle, presumably by inhibiting acid-mediated dissociation of the virion nucleic acid and proteins, which prevents nuclear transport of viral genome material.
Amantadine also prevents viral maturation in some strains of influenza A (for example, H7 strains) by promoting pH-induced conformational changes in influenza A haemagglutinin during intracellular transport late in the replication cycle. The virus's adsorption and penetration into cells do not appear to be affected by amantadine. In addition, amantadine does not interfere with the synthesis of viral components (for example, RNA-directed RNA polymerase activity).
Amantadine treatment of established influenza A infection does not appear to interfere with antibody response to the infection; however, some reduction in local immune responses has been seen in some patients. Because prophylactic use of amantadine can prevent influenza illness and, to a lesser extent, subclinical infection, some people who take amantadine can still develop immune responses that may protect them when they are exposed to the same or antigenically related viruses after stopping amantadine prophylaxis. Amantadine does not interfere with the immunogenicity of influenza A virus vaccine.
Amantadine-mediated increases in lysosomal pH may inhibit virus-induced membrane fusion in enveloped RNA viruses that are susceptible to higher concentrations of amantadine than those required to inhibit influenza A.
Pharmacokinetics
Absorption
Amantadine hydrochloride is well absorbed from the gastrointestinal tract, with peak blood concentrations occurring 1-4 hours after an oral dose. For a 100 mg capsule, average peak plasma concentrations are around 0.22 mcg/mL at 3.3 hours, and similar concentrations are seen with the oral solution. Higher doses can lead to more than proportional increases in plasma concentration, particularly above 200 mg daily.
Distribution
Amantadine is distributed into various tissues, including the heart, lungs and kidneys, with higher concentrations in lung tissue than in blood. It is also found in nasal secretions and breast milk, with a notable erythrocyte-to-plasma ratio. The volume of distribution is about 3-8 L/kg.
Elimination
The elimination half-life of amantadine averages about 24 hours. However, it can be prolonged in older patients and in those with renal impairment. It is mainly excreted unchanged in the urine, with some metabolites identified. Urine acidification can increase amantadine excretion, while haemodialysis removes only minimal amounts of the medicine.
Ingredients
The active ingredient in this medicine is amantadine hydrochloride. Each Symmetrel capsule contains 100 mg amantadine hydrochloride. The other ingredients are lactose, povidone, magnesium stearate, red iron oxide (E172), titanium dioxide (E171), gelatin and white printer ink.
Dosage
Symmetrel capsules are brownish-red hard gelatin capsules with SYMM printed on them in white. Symmetrel capsules come in packs of 56 capsules.
Amantadine hydrochloride is taken by mouth as a single daily dose or, preferably, in 2 equally divided doses to minimise temporary side effects. If insomnia occurs, the last daily dose should be taken several hours before bedtime.
| Population | Dosage |
|---|---|
| Adults (18-64 years) | 200 mg daily (single dose or 100 mg twice daily); maximum 100 mg daily for prophylaxis in some cases. |
| Older adults (65+ years) | 100 mg once daily; may need further reduction based on renal function. |
| Children (9-12 years) | 100 mg twice daily or 200 mg daily (in 1 or 2 divided doses). |
| Children (1-9 years) | 4.4-8.8 mg/kg daily (up to 150 mg max), given in 1 or 2 divided doses. |
| Children (<1 year) | Dosage not established; use must be determined by an Australian doctor. |
Duration of Therapy
Treatment
For symptomatic treatment of respiratory tract illness caused by influenza A virus, amantadine hydrochloride should be started as soon as possible, preferably within 24-48 hours after symptoms begin. The ACIP currently states that it may be advisable to stop amantadine treatment as soon as clinically appropriate, generally within 3-5 days or 24-48 hours after symptoms disappear, because there is some risk that strains of influenza A resistant to amantadine and rimantadine may emerge during treatment. However, immunocompromised people may need a longer course of treatment.
Prevention
When a presumed influenza A outbreak occurs in a hospital, nursing home or other institution housing high-risk patients, amantadine prophylaxis should be started as soon as possible after the outbreak is recognised and continued for at least 2 weeks or until about one week after the end of the outbreak. When amantadine hydrochloride is used as an adjunct to influenza virus vaccine, the medicine is usually given for 2 weeks after the vaccine to provide chemoprophylaxis until a protective antibody response develops. Children younger than 9 years receiving influenza virus vaccine for the first time may need amantadine prophylaxis for up to 6 weeks following vaccination or until 2 weeks after the second vaccine dose.
When vaccination is contraindicated, amantadine prophylaxis may be given throughout the local influenza A outbreak, which may last 6-12 weeks. Amantadine prophylaxis can also be given throughout the outbreak period when a poor antibody response to influenza vaccine is expected (for example, in patients with severe immunodeficiency, including acquired immunodeficiency syndrome (AIDS)).
The manufacturer states that prophylaxis with amantadine hydrochloride should be started before or as soon as possible after the patient has contact with someone who has a respiratory illness thought to be caused by influenza A virus, and should be continued for at least 10 days after a known exposure.
Dosage in Renal Impairment
In patients with renal impairment, the dose of amantadine hydrochloride should be adjusted carefully, and some clinicians recommend frequent monitoring of blood concentrations. One manufacturer recommends that patients with creatinine clearances of 15-50 mL/minute per 1.73 m2 receive 200 mg of amantadine on the first day, followed by 100 mg maintenance doses given once daily in patients with creatinine clearances of 30-50 mL/minute per 1.73 m2 or once every other day in those with creatinine clearances of 15-29 mL/minute per 1.73 m2. This manufacturer recommends that patients with creatinine clearances less than 15 mL/minute per 1.73 m2 and haemodialysis patients receive 200 mg of amantadine every 7 days.
Because dose adjustment based on creatinine clearance may provide only an approximation of the best dose for a given patient, these patients should be monitored so that side effects can be recognised promptly and either the dose can be reduced further or the medicine can be stopped if needed. Haemodialysis contributes minimally to the clearance of amantadine.
If You Forget to Take Symmetrel 100mg Capsules
If you miss a dose, take one as soon as you remember unless it is nearly time for your next dose. Then continue as usual. Do not take a double dose.
If You Stop Taking Symmetrel Capsules
Do not stop taking Symmetrel 100mg capsules suddenly, as your symptoms may get worse.
If you want to stop taking Symmetrel 100mg capsules, ask your doctor, who will tell you how to reduce the dose gradually.
If you are taking antidepressants (used to treat mental health conditions) and you suddenly stop taking Symmetrel capsules, you may develop symptoms including:
- fever;
- sweating;
- a rapid heartbeat;
- muscle stiffness (difficulty moving);
- loss of bladder control (you may have a sudden urge to pass urine).
Contact your doctor straight away if you develop any of these symptoms.
Some patients may notice that this medicine becomes less effective after taking it regularly for a few months. If you notice this, tell your doctor.
Ask your doctor or pharmacist if you have any further questions about using this product.

Important Safety Information
Take care when drinking alcohol while taking Symmetrel 100mg capsules, as it may affect you more than usual.
Symmetrel 100mg capsules should be taken with a glass of water.
Symmetrel 100mg capsules contain lactose. If your doctor has told you that you are intolerant to some sugars, contact your doctor before taking Symmetrel 100mg capsules.
Pregnancy and Breastfeeding
Do not take Symmetrel 100mg capsules if you are pregnant or trying to become pregnant. Do not take Symmetrel 100mg capsules if you are breastfeeding, because Symmetrel passes into breast milk and could harm your baby. Ask your doctor or pharmacist for advice before taking any medicine.
Driving and Using Machines
Taking Symmetrel capsules may cause blurred vision or make you feel dizzy. If this affects you, do not drive or use machines until the effect has worn off.
Resistant Strains of Influenza A Virus
Amantadine- and rimantadine-resistant strains of influenza A can develop in about 33% of patients treated with these antivirals. Patients may initially shed susceptible virus strains but can start shedding resistant strains after 2-7 days of treatment, with immunocompromised people potentially shedding them for longer.
To help limit resistance, amantadine treatment should be stopped after 3-5 days or once symptoms resolve. Although most patients recover even after resistant strains emerge, these strains are still pathogenic. They can reduce the effectiveness of prophylaxis for close contacts. People with influenza-like symptoms should avoid contact with uninfected individuals, regardless of antiviral treatment.
Pediatric Precautions
The safety and efficacy of amantadine in children younger than 1 year of age have not been established. When used in children, amantadine has caused central nervous system (CNS) symptoms, which resolved when the medicine was stopped. The incidence of CNS-related side effects appears to be higher in people receiving amantadine than in those receiving rimantadine. An increased incidence of seizures has been reported in children with an underlying seizure disorder receiving amantadine.
Geriatric Precautions
While the safety and efficacy of amantadine in older patients have not been explicitly established, the medicine has been used in many older patients. The frequency and severity of CNS side effects reported in people older than 65 years receiving amantadine are higher than those reported in older people receiving rimantadine. Older adults may have reduced renal function, and because people with renal impairment may be at increased risk of amantadine-induced toxicity, the dose of amantadine hydrochloride for adults in this age group should not exceed 100 mg daily. This dose may need to be reduced further in some older patients.
Mutagenicity and Carcinogenicity
Amantadine was not mutagenic in the Ames microbial test using Salmonella typhimurium or a mammalian mutagen assay using Chinese hamster ovary cells when the tests were performed with or without metabolic activation. In addition, there was no evidence of chromosome damage in an in vitro test using freshly derived and stimulated human peripheral blood lymphocytes (with or without metabolic activation) or an in vivo mouse bone marrow micronucleus test (140-550 mg/kg; estimated human equivalent dosage of 11.7-45.8 mg/kg based on body surface area conversion). Long-term animal studies have not been performed to evaluate the carcinogenic potential of amantadine.
HIV-Infected Individuals
In Australia, the Australian Immunisation Handbook recommends considering annual influenza vaccination for all people living with HIV aged 6 months or older, as they may be at increased risk of influenza-related complications.
Influenza virus is not traditionally classified as an opportunistic pathogen. However, many experts consider vaccination against the virus a logical step in any HIV-infected individual, whether symptomatic or asymptomatic, because of the possible risks of respiratory infections in these patients, and protective antibody levels are likely with many such vaccines.
Antiviral prophylaxis may be used alongside, or as an alternative to, influenza vaccination in people living with HIV who may have a poor antibody response to the vaccine and/or a high risk of exposure to influenza A, especially during influenza outbreaks or in institutional settings. In Australian clinical practice, oseltamivir may be used to help prevent influenza A and B infection, while rimantadine or amantadine may be considered for prevention of influenza A infection.
Contraindications
Amantadine should be used cautiously in patients with liver disease, uncontrolled psychosis, seizure disorders, or those taking CNS-active medicines. Patients with a history of seizures need close monitoring for increased seizure activity. Because of possible CNS effects and visual disturbances, amantadine may impair the ability to perform tasks requiring alertness, such as driving. It is contraindicated in people with untreated angle-closure glaucoma.
Neuroleptic malignant syndrome (NMS) has been reported, particularly when amantadine is reduced or withdrawn, so careful observation is needed in patients taking antipsychotics. Dose adjustments may be needed for those with renal impairment, congestive heart failure or orthostatic hypotension. Resistant strains of influenza A can emerge during treatment, posing a risk of transmission to high-risk individuals.
People with influenza-like illness should minimise contact with uninfected individuals. Clinicians should consider possible bacterial infections when treating suspected influenza cases. Amantadine is contraindicated in patients with known hypersensitivity to adamantane derivatives.
Do not take Symmetrel 100mg capsules in the following cases:
- if you are allergic (hypersensitive) to amantadine hydrochloride or any of the ingredients of Symmetrel capsules (see Section 6 Further information);
- if you have fits (convulsions), for example, epilepsy;
- if you have ever had an ulcer in your stomach or small intestine;
- if you have severe kidney disease;
- if you are pregnant or trying to become pregnant;
- if you are breastfeeding.
If any of the above applies to you, or if you need more information, speak to your doctor or pharmacist before taking Symmetrel capsules.
Before you take Symmetrel capsules, tell your doctor if:
- you have any liver or kidney disease;
- you have a history of disease affecting the heart and blood vessels;
- you are currently suffering from heart problems or heart failure (heart problems that cause shortness of breath or swollen ankles);
- you have any mental illness, for example, schizophrenia or dementia;
- you have increased pressure in the eyes (glaucoma).
If any of the above applies to you, or if you need more information, speak to your doctor or pharmacist before taking Symmetrel capsules.
Interactions
Tell your doctor or pharmacist if you are taking, or have recently taken, any of the following medicines, as they may interact with Symmetrel capsules:
| Anticholinergics | such as procyclidine, used to treat Parkinson's disease |
| Antispasmodics | such as hyoscine, used to treat stomach spasms or cramps |
| Levodopa | used to treat Parkinson's disease |
| Antipsychotics | such as chlorpromazine, haloperidol, used to improve thoughts, feelings and behaviour when these are disturbed in certain medical conditions |
| Diuretics | such as hydrochlorothiazide, amiloride, or triamterene, used to relieve fluid retention and reduce high blood pressure |
This medicine may also interact with the following:
- Influenza Virus Vaccine: Amantadine does not interfere with the antibody response to influenza vaccines and can be given at the same time.
- CNS Stimulants: Caution is advised when combining amantadine with CNS stimulants because of the risk of additive effects.
- Co-trimoxazole: Taking these together may reduce amantadine's renal clearance, mainly because of trimethoprim. Toxic delirium has been reported after this combination.
- Other Drugs: Combining amantadine with triamterene and hydrochlorothiazide increased plasma amantadine levels, although the specific cause is unclear. Quinidine or quinine may also reduce amantadine's renal clearance. In addition, using amantadine with CNS-active antihistamines can increase CNS side effects.
Please tell your doctor or Australian pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.

Side Effects
Amantadine can cause a range of side effects, which can be grouped by body system:
Common Side Effects
- Gastrointestinal: Nausea (5-10%), anorexia, constipation, diarrhoea, dry mouth (1-5%), and vomiting (up to 1%).
- Nervous System: Dizziness, insomnia, nervousness, confusion, agitation, hallucinations and psychotic episodes. Severe effects may include seizures and neuroleptic malignant syndrome on withdrawal.
- Cardiovascular: Orthostatic hypotension, peripheral oedema (1-5%), congestive heart failure, arrhythmias and tachycardia.
Less Common Side Effects
- Dermatological: Livedo reticularis (1-5%), rash and photosensitivity.
- Ocular: Visual disturbances such as corneal opacity and reduced visual acuity (up to 1%).
- Psychiatric: Increased risk of mania and suicidal ideation; symptoms may worsen in patients with a history of mental disorders.
Rare but Serious Effects
- Neurological: Peripheral neuropathy has been reported with long-term use; stopping treatment can lead to acute delirium.
- Haematological: Rare cases of leukopenia and neutropenia.
- Respiratory: Shortness of breath and, rarely, acute respiratory failure.
Side effects are often dose-related, with higher doses increasing the risk of severe reactions. People with kidney problems or a history of seizures are at greater risk of central nervous system effects. Regular monitoring is advised for people taking amantadine long term.
Toxicity
Manifestations
An overdose of amantadine can be fatal, with the lowest reported lethal dose being 2 g. Symptoms of acute overdose include heart problems (arrhythmias, tachycardia, hypertension), pulmonary oedema, kidney dysfunction, and central nervous system toxicity (insomnia, anxiety, confusion, hallucinations). Reported cases have included severe reactions such as seizures and hyperthermia. For example, one patient who took 2.5 g experienced coma and cardiopulmonary arrest.
Treatment
There is no specific antidote for amantadine overdose. If the overdose was recent, gastric lavage or induced vomiting may be needed. Supportive care includes monitoring vital signs and giving fluids. Electrocardiographic monitoring is essential because of the risk of tachyarrhythmias. Acidifying agents can increase amantadine excretion, but haemodialysis is only minimally effective. Close observation for hyperactivity and seizures is important; sedatives and anticonvulsants may be needed. Physostigmine has been used to manage central nervous system toxicity, but its risks should be taken into account.
Storage
Keep out of the reach and sight of children. Do not use Symmetrel capsules after the expiry date shown on the packaging. The expiry date refers to the last day of that month. If your doctor decides to stop your treatment, return any unused medicine to your pharmacist. Only keep it if your doctor tells you to.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines that are no longer needed. These measures will help protect the environment.