Valtrex (Valacyclovir)

Dosages
Valtrex 500 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | A$4.47 | A$134.09 | |
| 60 | A$3.78 | A$226.51 | |
| 90 | A$3.53 | A$317.64 | |
| 120 | A$3.43 | A$411.37 |
Valtrex 1000 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | A$7.33 | A$220.00 | |
| 60 | A$6.10 | A$365.81 | |
| 90 | A$5.70 | A$512.91 | |
| 120 | A$5.49 | A$658.71 |
Payment & Delivery
Your order is carefully packed and ships within 24 hours. Here is what a typical package looks like.
Sized like a regular personal letter (approximately 24x11x0.7 cm), with no indication of what is inside.



| Delivery Method | Estimated delivery |
|---|---|
| Express Free for orders over A$390.54 | Estimated delivery to Australia: 4-7 days |
| Standard Free for orders over A$260.36 | Estimated delivery to Australia: 14-21 days |










Discount Coupons
- Australia Day - 26 January 2026 10% AUSDAY10
- ANZAC Day - 25 April 2026 8% ANZAC8
- Boxing Day - 26 December 2026 12% BOXING12
Brand Names
| Country | Brand Names |
|---|---|
 Argentina Argentina | Viramixal Viranet |
 Belgium Belgium | Zelitrex |
 Denmark Denmark | Zelitrex |
 Finland Finland | Valavir |
 France France | Zelitrex |
 Italy Italy | Talavir Zelitrex |
 Mexico Mexico | Rapivir Valinir |
 Netherlands Netherlands | Zelitrex |
 Portugal Portugal | Crotax Valavir |
 Spain Spain | Valherpes Valpridol Virval |
| Manufacturer | Brand Names |
|---|---|
| Cipla Limited | Valcivir |
Description
What Valtrex is and what it is used for
Valtrex belongs to a group of medicines called antivirals. It works by killing or stopping the growth of viruses called herpes simplex virus (HSV), varicella zoster virus (VZV), and cytomegalovirus (CMV). In Australia, it is commonly prescribed in general practice and dermatology settings for these viral infections.
Valtrex can be used to:
- treat shingles (in adults)
- treat HSV infections of the skin and genital herpes (in adults and adolescents over 12 years old). It is also used to help prevent these infections from coming back.
- treat cold sores (in adults and adolescents over 12 years old)
- prevent infection with CMV after organ transplants (in adults and adolescents over 12 years old)
- treat and prevent HSV infections of the eye
Valtrex or acyclovir
| Comparison | Valtrex (valacyclovir) | Acyclovir |
|---|---|---|
| What it is | Prodrug that converts to aciclovir in the body. | Active antiviral medicine (parent compound). |
| Absorption / bioavailability | Higher oral bioavailability than oral aciclovir (often allowing less frequent dosing). | Lower oral bioavailability; dosing tends to be more frequent. |
| Dosing convenience (common HSV use) | Often once daily for suppression; short-course regimens are usually simpler. | Often 2+ times daily for suppression; short-course regimens can be more frequent. |
| Suppressive therapy (recurrent genital HSV, typical) | 1 g once daily, or 500 mg once daily for some patients (per labelling). | 400 mg twice daily for up to 12 months, then re-evaluate (per labelling). |
| Reducing HSV-2 transmission (evidence) | 500 mg once daily can reduce HSV-2 transmission in discordant heterosexual couples as part of a prevention strategy. | Used for suppression, but the commonly cited transmission-reduction regimen is for valacyclovir. |
| Available dosage forms | Mainly oral tablets/caplets. | Multiple forms exist (including oral; some products also come in topical and IV forms). |
| Kidney considerations | Renal dose adjustments may be needed; take extra care if you have kidney disease or take nephrotoxic medicines. | Renal dose adjustments may be needed; take extra care if you have kidney disease or take nephrotoxic medicines. |
| Common side effects | Often mild (for example, headache or nausea), depending on dose and individual tolerance. | Often mild (for example, headache or nausea), depending on dose and individual tolerance. |
| Cost / generics | Generic valacyclovir is widely available; branded Valtrex is usually more expensive. | Generic aciclovir is widely available and often cheaper. |
| Bottom line | Often chosen for convenience (fewer daily doses). | Often chosen for cost and the wider range of formulations. |
Before you take Valtrex
Don't take Valtrex if you are allergic (hypersensitive) to valaciclovir or aciclovir, or any of the other ingredients.
Don't take Valtrex if this applies to you. If you are not sure, talk to your doctor or pharmacist before taking Valtrex.

Take special care with Valtrex
Check with your doctor or pharmacist before taking Valtrex oral tablets 250 mg, 500 mg, or 1000 mg if:
- you have kidney problems
- you have liver problems
- you are over 65 years of age
- your immune system is weak
If you are not sure whether any of the above apply to you, talk to your doctor or pharmacist before taking Valtrex 250 mg, 500 mg, or 1000 mg film-coated tablets.
Prevent passing genital herpes on to others
If you are taking Valtrex to treat or prevent genital herpes, or you have had genital herpes in the past, you should still practise safe sex, including using condoms. This is important to help prevent passing the infection on to others. You should not have sex if you have genital sores or blisters.
Taking other medications
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines. This includes medicines obtained without a prescription, including herbal medicines.
Tell your doctor or pharmacist if you are taking any other medicines that affect the kidneys. These include: aminoglycosides, organoplatinum compounds, iodinated contrast media, methotrexate, pentamidine, foscarnet, ciclosporin, tacrolimus, cimetidine and probenecid.
Always tell your doctor or pharmacist about other medicines if you are taking Valtrex to treat shingles or after having an organ transplant.
Pregnancy and breast-feeding
Valtrex is not usually recommended during pregnancy. If you are pregnant, think you may be pregnant, or are planning to become pregnant, don't take Valtrex without checking with your doctor. In Australia, your doctor will weigh up the benefit to you against the risk to your baby of taking Valtrex while you're pregnant or breastfeeding.
Driving or using machines
Valtrex can cause side effects that may affect your ability to drive. Don't drive or use machines unless you are sure you are not affected.
How to take Valtrex oral tablets
Always take Valtrex exactly as your doctor has told you. If you are in Australia, check with your doctor or pharmacist if you are not sure.
The dose you need will depend on why your doctor has prescribed Valtrex for you. Your doctor will discuss this with you.
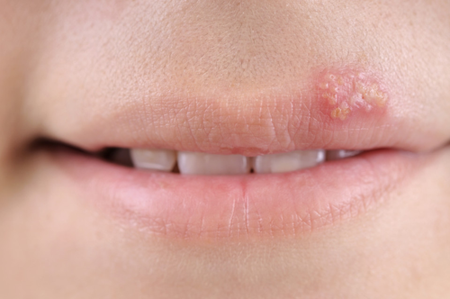
Treatment of shingles
The usual dose is 1000 mg (one 1000 mg tablet or two 500 mg tablets) three times a day.
You should take Valtrex for seven days.
Treatment of cold sores
The usual dose is 2000 mg (two 1000 mg tablets or four 500 mg tablets) twice a day.
The second dose should be taken 12 hours (no sooner than 6 hours) after the first dose.
You should take Valtrex for one day only (two doses).
Treatment of HSV infections of the skin and genital herpes
The usual dose is 500 mg (one 500 mg tablet or two 250 mg tablets) twice a day.
For the first infection, you should take Valtrex for five days, or for up to ten days if your doctor tells you to. For recurrent infection, the treatment duration is usually 3-5 days.
Helping to prevent HSV infections from returning after you have had them
The usual dose is one 500 mg tablet once a day.
Some people with frequent recurrences may benefit from taking one 250 mg tablet twice a day.
You should take Valtrex until your doctor tells you to stop.
To stop you being infected with CMV (Cytomegalovirus)
The usual dose is 2000 mg (two 1000 mg tablets or four 500 mg tablets) four times a day.
You should take each dose about 6 hours apart.
You will usually start taking Valtrex as soon as possible after your surgery.
You should take Valtrex for around 90 days after your surgery, until your doctor tells you to stop.
Your doctor may adjust the dose of Valtrex if:
- you are over 65 years of age
- you have a weak immune system
- you have kidney problems.
Talk to your doctor before taking Valtrex if any of the above apply.
Taking this medication
Take this medicine by mouth.
Swallow the tablets whole with a drink of water.
Take Valtrex at the same time each day.
Take Valtrex according to instructions from your Australian doctor or pharmacist.
People over 65 years of age or those with kidney problems: while you are taking Valtrex, it is very important to drink water regularly during the day. This will help reduce side effects that can affect the kidneys or nervous system. Your doctor will monitor you closely for signs of these effects. Nervous system side effects may include feeling confused or agitated, or feeling unusually sleepy or drowsy.
If you take more Valtrex than you should
Valtrex is not usually harmful unless you take too much over several days. If you take too many tablets, you may feel sick, vomit, or become confused, agitated, or unusually sleepy. Talk to your doctor or pharmacist if you take too much Valtrex. Take the medicine pack with you.
If you forget to take Valtrex
If you forget to take Valtrex, take it as soon as you remember. However, if it is nearly time for your next dose, skip the missed dose.
Don't take a double dose to make up for a forgotten dose.
| Pros of taking Valtrex daily | Cons / downsides |
|---|---|
| May reduce the number of HSV outbreaks (and make them milder for many people). | Does not eliminate HSV transmission risk; asymptomatic shedding can still occur. |
| Can lower the risk of passing HSV-2 to a partner when used as part of a prevention plan. | Possible side effects, most commonly headache or nausea (varies by person). |
| Convenient suppressive dosing options (often once daily). | Kidney-related considerations: dose adjustments may be needed with reduced kidney function; take extra care with other nephrotoxic medicines. |
| May improve quality of life by reducing recurrences and anxiety about flare-ups. | Long-term use: labelling notes limited evidence for suppressive therapy beyond 1 year in immunocompetent patients. |
| May reduce time spent dealing with episodic treatment and symptoms. | Daily tablet trade-offs (cost, adherence, and personal preference for episodic versus suppressive therapy). |
| Antiviral resistance is uncommon in immunocompetent people. | Resistance is still possible, especially in immunocompromised patients (rare overall). |
Valaciclovir: Organs and Systems
Nervous system
Valaciclovir is a prodrug of aciclovir and can therefore cause similar effects, as two cases of nervous system effects have shown.
- A 65-year-old man was given valaciclovir 1 g twice daily for 36 hours and had reduced concentration and was incoherent. All investigations were normal or negative. He improved rapidly after valaciclovir was stopped.
- A 44-year-old man was given valaciclovir 1 g three times daily for 5 days and developed a fever, disorientation, confusion, ataxia, dysarthria, and photophobia. All investigations were normal or negative. He was given antimicrobial medicines, including aciclovir, but his symptoms did not improve until the aciclovir was stopped.
Elderly patients and people with chronic renal insufficiency are most susceptible to the neurotoxic effects of aciclovir: confusion, hallucinations, dizziness, irritability, ataxia, tremor, myoclonus, and seizures. Symptoms usually occur within 3 days of starting treatment and resolve within 5 days after stopping it. Plasma aciclovir concentrations do not correlate with symptoms. Lumbar puncture and CT scans of the head are essentially unremarkable. The most common electroencephalographic abnormality is diffuse generalised slowing of brain wave activity.
Psychological, psychiatric
At high doses (8 g/day), hallucinations and confusion were a significant concern, but similar symptoms have also occurred at lower doses and in patients with renal insufficiency.
Visual and auditory hallucinations have been reported in a 60-year-old woman on CAPD.
A 58-year-old man with chronic renal insufficiency, who was haemodialysed twice a week, was treated with valaciclovir (1 g three times daily) for Herpes zoster. Two days later, he became disoriented, dizzy, dysarthric, and experienced hallucinations. The serum aciclovir concentration was 21 µg/ml. Treatment was stopped and he was treated with haemodialysis for 6 hours, resulting in marked clinical improvement. The next day, his symptoms of dysarthria recurred but immediately and completely resolved after a second haemodialysis.
Hematologic
In one study, high-dose valaciclovir was associated with an increased risk of a thrombotic microangiopathy-like syndrome, reported as thrombocytopenic purpura or haemolytic-uraemic syndrome. This syndrome occurred in 14 of 523 patients who received valaciclovir and in only four of 704 patients who received aciclovir after a median of 54 (range) weeks of treatment.
The precise relation to valaciclovir remains unclear, since eight of 14 patients who were treated with valaciclovir had stopped treatment at least 1 week before the onset of the syndrome. In addition, all patients with thrombotic microangiopathy-like syndromes had taken multiple concomitant medicines, and most had other intercurrent illnesses, which could have explained the haematological and renal abnormalities. The authors concluded that additional data are required to understand the role of valaciclovir and other medicines in thrombotic microangiopathy-like syndromes, which are being recognised with increasing frequency in patients with advanced HIV disease.
Gastrointestinal
Nausea, vomiting, and abdominal pain were commonly reported in human volunteers, but only diarrhoea was significantly associated with exposure.
Urinary tract
Aciclovir is excreted by the kidneys. High plasma concentrations of aciclovir can lead to precipitation in the renal tubules, causing impaired renal function, which is generally reversible. Since oral valaciclovir can result in plasma aciclovir concentrations comparable to those reached with intravenous dosing, reversible impairment of renal function can also occur after prolonged use of high-dose valaciclovir. In a study of high-dose valaciclovir for prevention of cytomegalovirus disease in people with HIV, there was an association between treatment with valaciclovir and moderate nephrotoxicity (serum creatinine more than 1.5 times the upper limit of normal; estimated creatinine clearance under 50 ml/minute).
Valaciclovir: Side Effects
Valaciclovir is the L-valyl ester of aciclovir. After oral administration, it is rapidly and extensively converted to aciclovir by first-pass metabolism, resulting in plasma aciclovir concentrations previously only attainable with intravenous administration. Like aciclovir, valaciclovir is generally well tolerated. Compared with oral aciclovir, the systemic availability of aciclovir from oral valaciclovir is markedly improved.
Valaciclovir is highly active against Herpes simplex and Herpes zoster. It is also effective in suppressing recurrent episodes of genital herpes. Prophylactic administration of high doses of valaciclovir to prevent CMV disease was effective in patients with AIDS and in liver transplant recipients.
Observational studies
In a double-blind comparison of two regimens of valaciclovir 500 mg twice daily for recurrent genital herpes, a 5-day course and a 3-day course, there were no significant differences in therapeutic outcome or adverse events between the two regimens. The most common side effects were headache (10%), nausea (4%), diarrhoea (3%), and fatigue (1.5%).
Comparative studies
The effects of aciclovir and valaciclovir for anogenital herpes have been studied in people with HIV in two controlled trials. In the first study, 1062 patients with CD4+ counts over 100 x 10^6/l received valaciclovir or aciclovir for 1 year and were assessed monthly. In the second study, 467 patients were treated episodically for at least 5 days with valaciclovir or aciclovir and were assessed daily. Valaciclovir was as effective as aciclovir for suppression and episodic treatment of herpesvirus infections. Hazard ratios for the time to recurrence with valaciclovir 500 mg twice daily and 1000 mg once daily compared with aciclovir were 0.73 (95% CI = 0.50) and 1.31 (0.94). Valaciclovir 1000 mg twice daily and aciclovir had similar effects on the duration of infective episodes (HR - 0.92; CI = 0.75). The most common side effects, which occurred at similar rates with all regimens, were diarrhoea, headache, infections, rashes, nausea, rhinitis, pharyngitis, abdominal pain, fever, depression, and cough.
Placebo-controlled studies
In large, placebo-controlled comparisons of the efficacy of valaciclovir and aciclovir in treating or suppressing recurrent genital Herpes simplex infections in immunocompetent people, dosages up to 2 g/day were well tolerated, with safety profiles comparable to aciclovir. In a comparison of high-dose valaciclovir (8 g/day) with two doses of aciclovir (0.8 and 3.2 g/day) for prophylaxis of cytomegalovirus disease in patients with advanced human immunodeficiency virus infection, intention-to-treat analysis showed a trend towards earlier mortality in those who received valaciclovir. In those who actually received valaciclovir, survival was significantly shorter. In view of the unexplained trend towards earlier mortality, as well as higher frequencies of renal toxicity and premature treatment discontinuation, the authors concluded that the dose of valaciclovir was too high and that better tolerated doses, which maintain a protective effect on cytomegalovirus disease, need to be identified.
Second-Generation Effects
Fetotoxicity
In a phase I trial, valaciclovir administered in the third trimester of pregnancy was well tolerated.
Susceptibility Factors
Renal disease
Side effects of valaciclovir, the L-valyl ester of aciclovir, can be associated with increased drug concentrations when the dose is not adjusted for reduced renal function. For example, aseptic meningitis has been associated with valaciclovir in a patient with renal insufficiency.
An 88-year-old man with renal insufficiency took valaciclovir 1000 mg three times daily. After the first dose, he became disoriented and incontinent. Valaciclovir was stopped, but the symptoms continued and progressed to drowsiness and nuchal rigidity. After an extensive work-up, aseptic meningitis was diagnosed.
Given the patient's age and renal dysfunction, it is likely that excessive valaciclovir accumulation was responsible for this presentation.
Drug-Drug Interactions
Cimetidine
In an open, single-dose study of the effects of probenecid and cimetidine on the pharmacokinetics of valaciclovir and its metabolite aciclovir in 12 healthy men, valaciclovir 1 g, valaciclovir plus probenecid 1 g, valaciclovir plus cimetidine 800 mg, and valaciclovir with a combination of probenecid and cimetidine were studied. At three subsequent administrations, drug regimens were alternated among groups so that each group received each regimen. Probenecid and cimetidine respectively increased the mean Cmax of valaciclovir by 23 and 53% and its AUC by 22 and 73%. Probenecid and cimetidine also respectively increased the mean aciclovir Cmax by 22 and 8% and its AUC by 48 and 27%. The combination had a greater effect than either drug alone. Neither cimetidine nor probenecid affected the absorption of valaciclovir.
Probenecid
In an open, single-dose study of the effects of probenecid and cimetidine on the pharmacokinetics of valaciclovir and its metabolite aciclovir in 12 healthy men, valaciclovir 1 g, valaciclovir plus probenecid 1 g, valaciclovir plus cimetidine 800 mg, and valaciclovir with a combination of probenecid and cimetidine were studied. At three subsequent administrations, drug regimens were alternated among groups so that each group received each regimen. Probenecid and cimetidine respectively increased the mean Cmax of valaciclovir by 23 and 53% and its AUC by 22 and 73%. Probenecid and cimetidine also respectively increased the mean aciclovir Cmax by 22 and 8% and its AUC by 48 and 27%. The combination had a greater effect than either drug alone. Neither cimetidine nor probenecid affected the absorption of valaciclovir.
How to store
Keep out of the reach and sight of children.
Do not use Valtrex after the expiry date stated on the carton. The expiry date (Exp.) refers to the last day of that month.
Store below 30°C (86°F).
Medicines should not be disposed of via wastewater or household waste. In Australia, ask your pharmacist how to dispose of medicines you no longer need. This will help protect the environment.